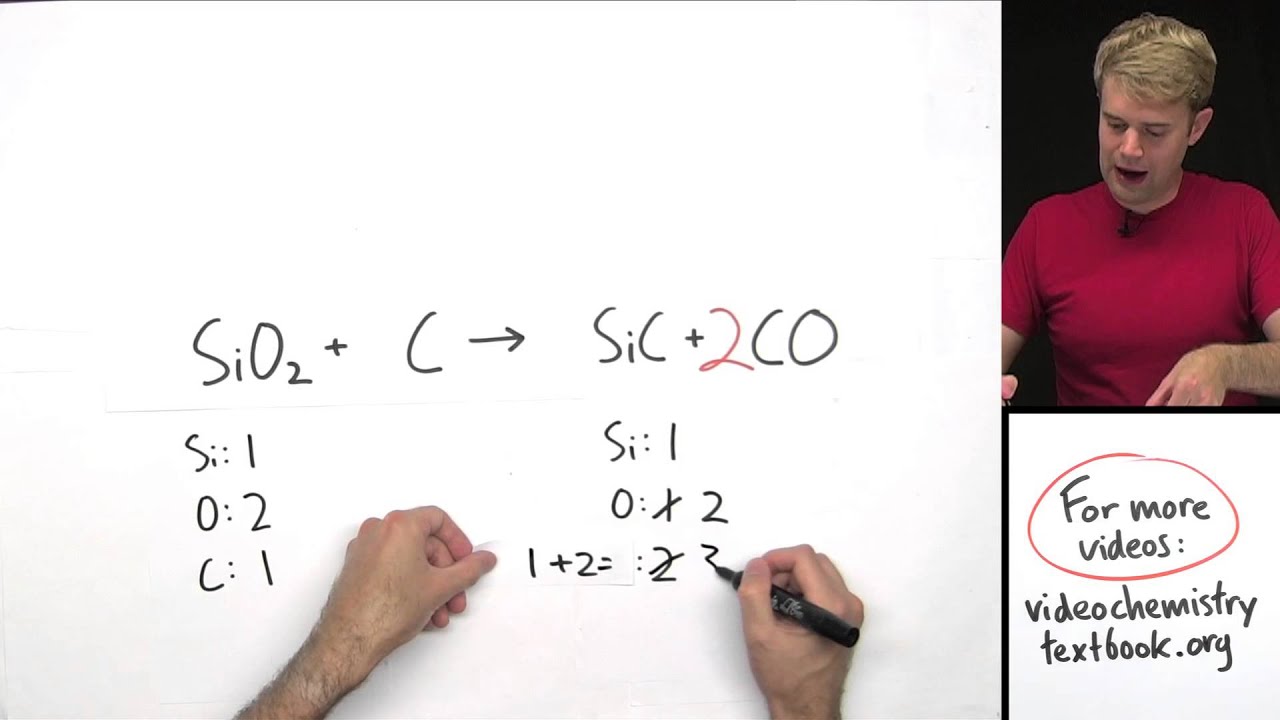 
Multiply the two half-reactions by the number that yields the lowest common number of electrons in each half-reaction.MnO 4 - + 8H + + 5e - → Mn 2+ + 4H 2O 2I - → I 2 + 2e - Equations Inequalities Scientific Calculator Scientific Notation Arithmetics Complex Numbers Polar/Cartesian Simultaneous Equations System of Inequalities Polynomials Rationales Functions Arithmetic & Comp. The second half-reaction has 2- on the left and 0 on the right. Add 5 electrons to the left to balance the charge. In this example, the first half-reaction has a charge of 7+ on the left and 2+ on the right. The Stoichiometry Calculator is used to balance the chemical equations on both sides. Now, balance charge by adding electrons as needed. Stoichiometry Calculator + Online Solver With Free Steps.Balance the hydrogen by adding H + as needed: MnO 4 - + 8H + → Mn 2+ + 4H 2O 2I - → I 2.Compare your results with real-life examples and simulations of chemical reactions. Practice with different levels of difficulty and get immediate feedback. Now add O and H 2O as needed to balance oxygen: MnO 4 - → Mn 2+ + 4H 2O 2I - → I 2 Learn how to balance chemical equations by using the law of conservation of mass and the coefficients of reactants and products.Balance all of the atoms besides the oxygen (O) and hydrogen (H) in the half-reactions: MnO4 - → Mn 2+ 2I - → I 2.Write a skeleton ionic equation that only covers the atoms that change oxidation number: MnO 4 - → Mn 2+ I - → I 2.Find the atoms that experience a change in oxidation number: Mn: +7 → +2 I: +1 → 0.Write down the oxidation numbers for each type of atom on both sides of the equation: Left hand side: K = +1 Mn = +7 O = -2 I = 0 H = +1 S = +6 Right hand side: I = 0 Mn = +2, S = +6 O = -2.First, write the unbalanced chemical equation: KMnO 4 + KI + H2SO 4 → I 2 + MnSO 4.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
